
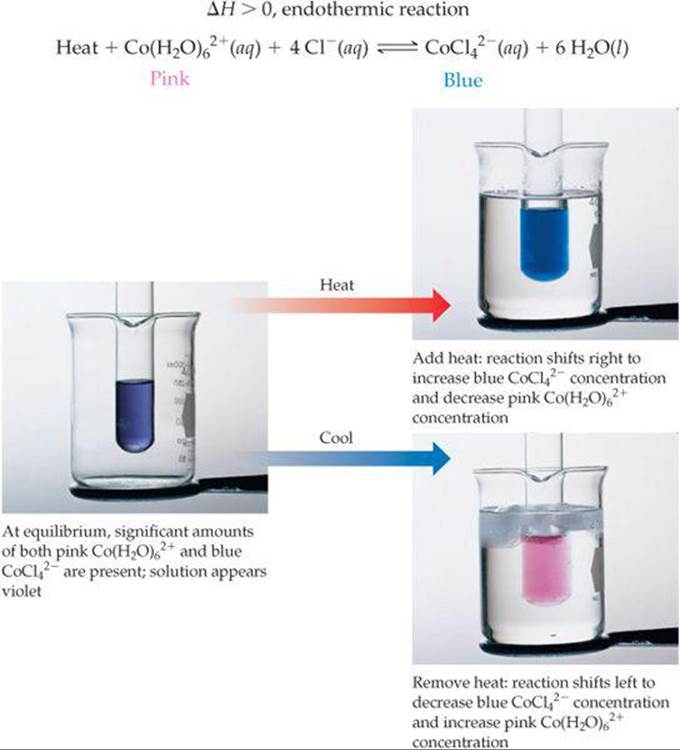
Step 2: Add AgNO₃, then measure and calculate Step 1: Add HCl, then measure and calculateĭefine the reaction being studied (leave blank if it balanced chemical Watch the following video for see what the equilibrium looks like in a real lab setting. The cobalt(II) ion bound to water is a pink color where as the cobalt(II) ion bound to chloride ions is blue.Ĭ o ( H 2 O ) 2 + 6 ( a q ) + 4 C l − ( a q ) ⇀ ↽ C o C l 2 − 4 ( a q ) + 6 H 2 O ( l ) C o ( H 2 O ) 6 2 + ( a q ) + 4 C l − ( a q ) ↽ ⇀ C o C l 4 2 − ( a q ) + 6 H 2 O ( l ) In this simulation, we will examine the reaction between hexaaquacobalt(II) ion in equilibrium with tetrachlorocobaltate(II) ion. These complexes often exhibit different colors, depending on what is bound. Many transition metal ions can form bonds with electron rich compounds or ions. The reaction examined will use something called a complex ion. In this section, you will examine an equilibrium reaction that occurs in solution. Solution Equilibria: Cobalt(II) complex ions Results were adequately recorded and calculated correctly. This criterion is linked to a Learning Outcome Analysis and FormatDiscussion is written in complete sentences and in paragraph form. Uses quantitative experimental results to explain temperature affects the equilibrium constant for an endothermic reaction? (or how does temperature affect the equilibrium constant for an exothermic reaction?) This criterion is linked to a Learning Outcome Discussion: Effect of temperature on KcUses quantitative experimental results to explain whether hanging the temperature of a reaction change the equilibrium constant? This criterion is linked to a Learning Outcome Discussion: KcUses quantitative experimental results to explain whether changing the concentration of reactant or product change the equilibrium constant for a reaction? 
This criterion is linked to a Learning Outcome Discussion: Endothermic or exothermicUses description of results to explain whether the reaction (as written in the Background) endothermic or exothermic. You need to conduct the experiment on the ChemCollective simulator, so I have blurred out some sections in the video.ĭiscussion: Color changesUses experimental observations to explain whether color changes observed in steps 1-4 consistent with Le Chatelier’s Principle? This file is downloadable: cobalt_BYH.pdf downloadįor help with ChemCollective and with the Excel sheet, you can view the video below. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
